eCOA


The AI Pilot Trap and How Clinical Trial Leaders Can Escape It
Most AI pilots in clinical trials fail to scale beyond proof of concept. Learn practical strategies for moving from isolated experiments to enterprise adoption.



The 1:1:1 vision: Reimagining clinical development
"The scarcest resource in clinical trials is the time of the highly qualified people running the clinical trials. We need to free up their time to bring more meaningful innovation to patients."
- David Hyman, Chief Medical Officer, Eli Lilly
Since the year 2000, the pace of drug approvals has remained stubbornly slow, with the FDA approving roughly 50 new treatments per year. This pace is in spite of massive increases in R&D investment. It’s well known that clinical trials take 10-12 years on average to complete all four phases. But what if clinical trials didn’t take weeks to start, months to enroll, and years to complete?


Case study: Scaling global vaccine mega-trials for a top-5 pharma
Learn how Medable enabled a top-5 pharma to scale vaccine mega-trials with near-100% enrollment, real-time safety data, and >90% diary compliance.


From the Congress floor to the dinner table: A Week in Washington that reminded me why this work matters
There's a particular kind of energy at the World Vaccine Congress that's hard to describe unless you've been in it. Hundreds of scientists, executives, policymakers, pharma leaders, CRO teams, and site networks, all in one convention centre, all sitting with the same fundamental tension. We know how to make vaccines that save lives. So why does getting them to patients still take so long?
I spent three days in Washington this week as part of the Medable team. Washington in late March meant that the cherry blossoms were just past their peak but still stunning, and catching them along the Tidal Basin between sessions was one of those small, unexpected gifts that a busy conference week doesn't always make room for.
For the conference, I came in with a clear intention. I wanted to reconnect with partners I respect, listen more than I talk, and have honest conversations about where this industry is and where it needs to go. What I didn't fully anticipate was how much the week would reinforce something I already believed but needed reminding of.
The urgency is real. And it's shared.


Case study: ICON and Medable drive 85% eConsent adoption in U.S. menopause study
How do you drive adoption in a complex women’s health study? See how ICON and Medable reached 85% eConsent uptake across 1,200+ participants with a smarter, site-first approach.


Harnessing AI for more efficient clinical trials
Explore how AI is transforming clinical trials, from accelerating data analysis to predicting trial outcomes.


eCOA vs ePRO: Understanding the differences in clinical trials
Digital data capture has become essential to modern clinical research. Sponsors and research organizations increasingly rely on electronic outcome assessment tools to collect high quality patient data, reduce manual errors, and improve regulatory compliance.
Two terms appear frequently in this space: eCOA (electronic Clinical Outcome Assessment) and ePRO (electronic Patient Reported Outcome).
These terms are closely related. However, they are not interchangeable.


Everest analysis: How Medable eCOA solves speed, patient experience, and customer needs
eCOA has moved from a supporting tool to a foundational pillar of modern clinical trials, and Everest Group agrees. In its inaugural eCOA Products PEAK Matrix Assessment, Everest named Medable a Leader, citing strong market impact, accelerated timelines, and a platform built for real-world trial complexity. As the eCOA market surges toward nearly $1B in value, this recognition underscores how speed, patient experience, and AI-driven innovation are reshaping how trials are designed, launched, and scaled globally.


Playing catch-up: FDA wants “patient’s voice” ePRO in your oncology trial
For years now, the FDA has been making one point crystal clear to sponsors and CROs across our industry; they want the patient voice incorporated whenever possible in oncology trials.
The FDA's initiative is driven by the recognition that a patient's personal experience with a disease and its treatment is a unique and essential measure of a medical product's benefit and risk.
- Rationale: The FDA explicitly states that "patients provide a unique perspective on treatment effectiveness" and "some treatment effects are known only to the patient." Outcomes that truly matter to patients, such as functioning, quality of life, and the burden of side effects, are often best measured directly by the patient.
- Mandate: The Patient-Focused Drug Development (PFDD) effort, codified in part by the 21st Century Cures Act, requires the inclusion of such patient experience data in clinical research.
- Guidance series: To formalize this approach, the FDA has released a series of methodological guidance documents (the PFDD Guidance Series) that outline how stakeholders should collect, submit, and use patient input to inform medical product development.


Medable oncology solutions
Discover how Medable’s AI-powered oncology platform simplifies complex cancer trials by integrating eCOA, ePRO, and eConsent solutions—reducing trial time, improving patient retention, and enhancing data quality for faster, more efficient research.


Six steps to help you choose the right clinical trial partner
“This year, we’ve decided to stick to paper. We know what we’re getting and we’ve always done it this way.”
That was the unwavering response from a clinical operations lead at a pioneering biotech when asked why they still relied on paper diaries for patient-reported outcomes.
Despite the growing complexity of global trials, the promise of real-time data, and the surge of digital capabilities available today, some organizations have held tight to a method of clinical trial conduct that’s increasingly as outclassed as it is outdated.
It’s well known that individuals and organizations believe that change can be daunting, stressful, and difficult, especially when the old way is familiar and entrenched. However, much like anything else, having a partner who can help guide you through the process is massively important. But, how do you find the right partner for your trials?
Before locking in a vendor, organizations must engage in a deliberate process to assess their needs, align stakeholders, and set the foundation for long-term success. This blog explores the critical steps sponsors should take before selecting a digital partner, using insights and frameworks drawn from Medable’s therapeutic area standards and industry best practices.
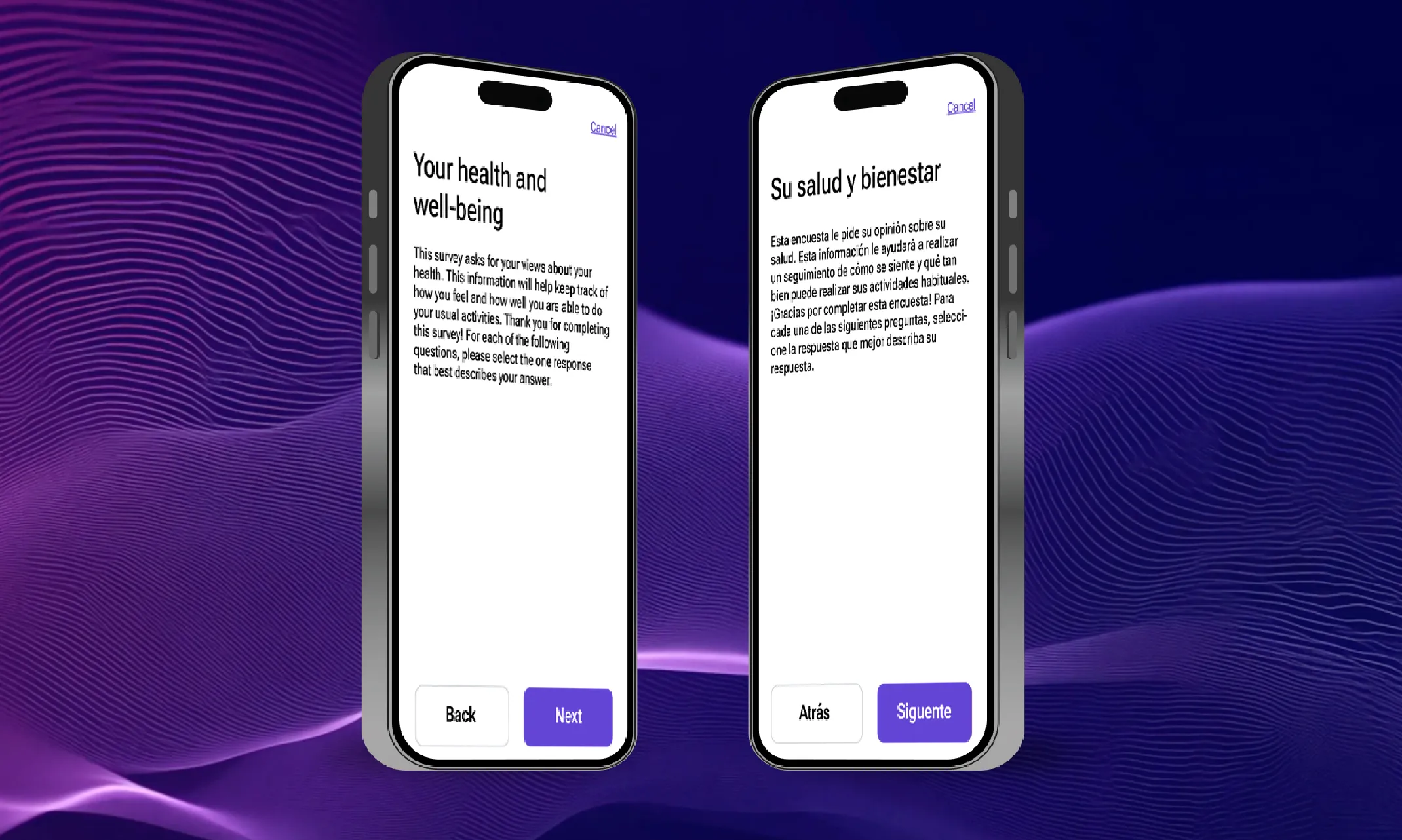

From bottlenecks to breakthroughs: How AI is transforming translation timelines
According to ClinicalTrials.gov, there are 3,046 multi-country trials being conducted this year. While many trials remain localized within a single country, there has been a definitive movement towards conducting trials in multiple countries, especially for larger, later-stage trials. This is driven by the positives that multi-country trials offer, like faster patient recruitment, lower costs in some regions, and the need for diverse patient populations.. However, behind the scenes, a critical bottleneck has been slowing many trials down. This bottleneck is the translation process that’s required to make trials work across multiple languages, locales, and regulatory bodies/organizations.
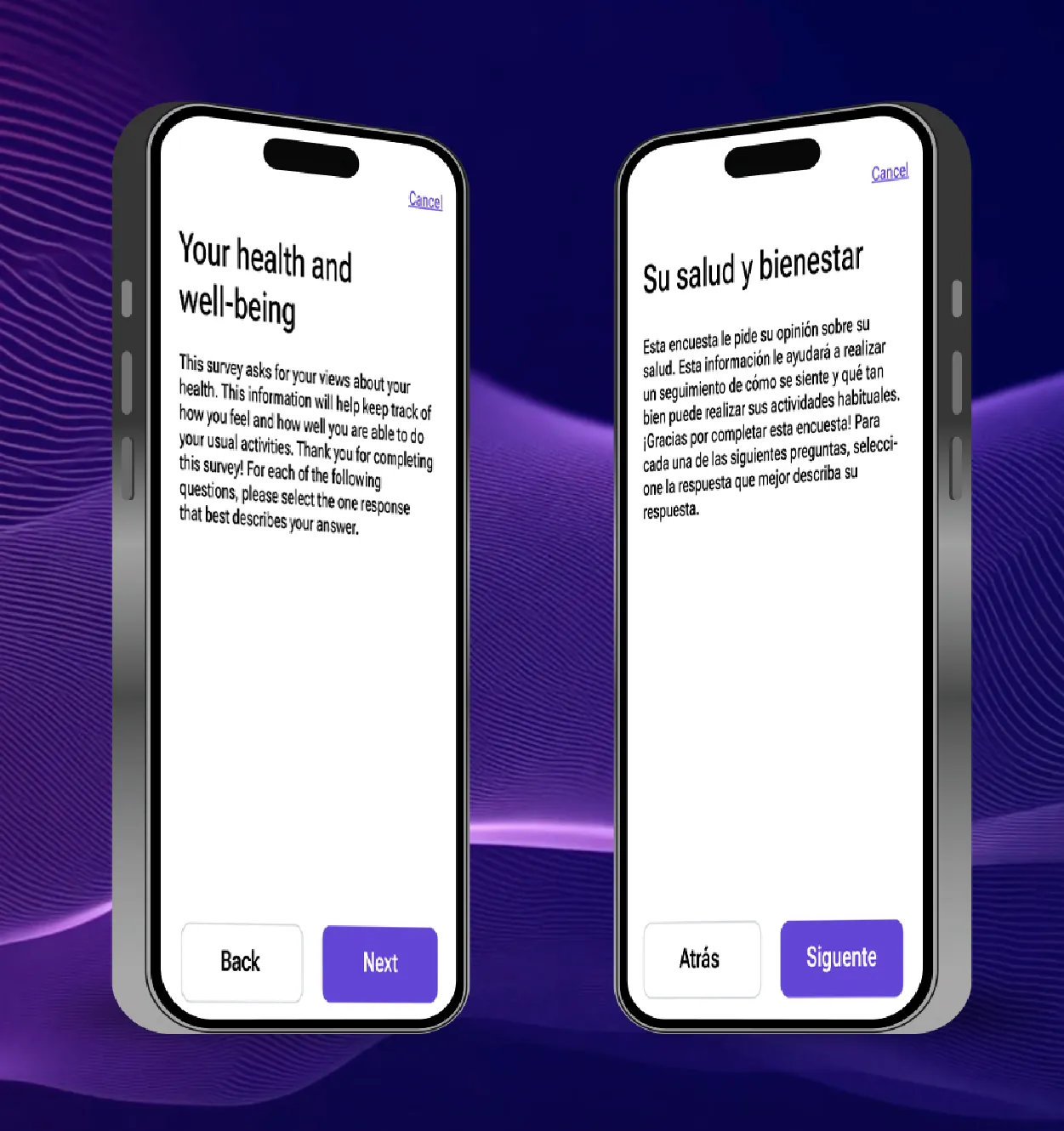

Case study: Removing translation bottlenecks with AI
Traditionally, translations and language migration create significant bottlenecks on the path to trial study go-live. The process is traditionally manual, linear, and resource intensive. To address these challenges, Medable partnered with Lionbridge, a leading translation services company to compare the status quo translation process against an AI-enabled approach powered by both companies’ proprietary new AI tools.



